Just as is observed with proteins in nature, additional structural diversity can be imparted to glycans by modifying their hydroxyl groups with phosphate, sulfate, or acetyl esters, and/or their amino groups with acetyl or sulfate groups.Ī carbohydrate may be termed “complex” if it contains more than one type of monosaccharide building unit. The prefix “glyco” and the suffixes “saccharide” and “glycan” indicate the presence of carbohydrate constituents (e.g., glycoproteins, glycolipids, and proteoglycans). The term “glycoconjugate” is often used to describe a macromolecule that contains monosaccharides covalently linked to proteins or lipids. Thus, the relationship of monosaccharides to oligosaccharides or polysaccharides is analogous to that of amino acids and proteins or nucleotides and nucleic acids (polynucleotides). The term “polysaccharide” is typically used to denote any linear or branched polymer consisting of monosaccharide residues, such as cellulose ( Chapters 14 and 24). Typically, the term “oligosaccharide” refers to any glycan that contains less than 20 monosaccharide residues connected by glycosidic linkages. Monosaccharides are joined together to give rise to oligosaccharides or polysaccharides. Monosaccharides are the simplest of these polyhydroxylated carbonyl compounds (saccharide is derived from the Greek word for sugar or sweetness). X(H 2O) n that also possess a carbonyl group, either an aldehyde or a ketone. In the 19th century, sugar-based substances were referred to as carbohydrates, or “hydrates of carbon,” that are based on the general formula C Still, a host of names are commonly used to refer to sugar polymers in other textbooks and the literature. Acetal derivatives have been prepared by acid-catalyzed reactions with benzaldehyde and acetone.In this book, as well as in the earlier editions, the term glycan is used. A pyranose structure for D-glucose is drawn in the rose-shaded box on the left. The formation of acetal derivatives illustrates how subtle changes may alter this selectivity. Aldolhexoses usually form pyranose rings and their pentose homologs tend to prefer the furanose form, but there are many counter examples. The size of the cyclic hemiacetal ring adopted by a given sugar is not constant, but may vary with substituents and other structural features. Examples of four typical pyranose structures are shown below, both as Haworth projections and as the more representative chair conformers. 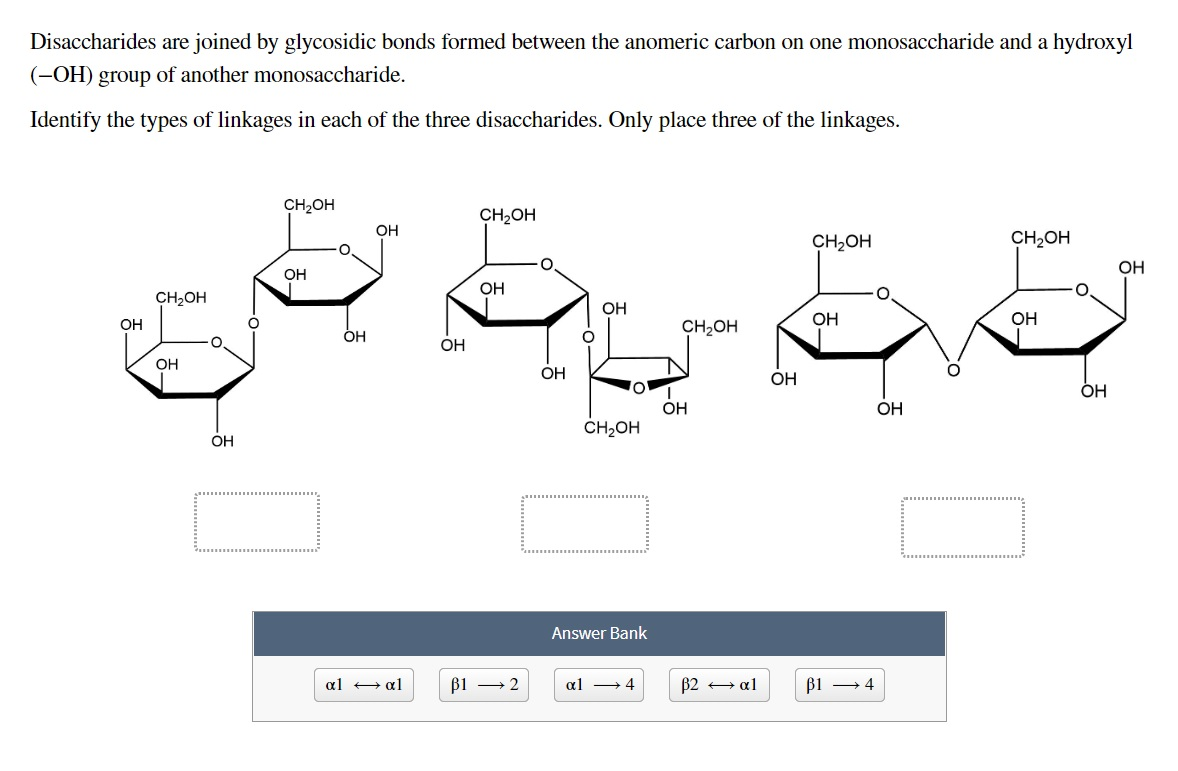
We know that these molecules are actually puckered in a fashion we call a chair conformation. These Haworth formulas are convenient for displaying stereochemical relationships, but do not represent the true shape of the molecules. In the D-family, the alpha and beta bonds have the same orientation defined for the furanose ring (beta is up & alpha is down).  
As with the furanose ring, the anomeric carbon is placed on the right with the ring oxygen to the back of the edgewise view. The cyclic pyranose forms of various monosaccharides are often drawn in a flat projection known as a Haworth formula, after the British chemist, Norman Haworth. The upper bond to this carbon is defined as beta, the lower bond then is alpha. The anomeric carbon atom (colored red here) is placed on the right. By convention for the D-family, the five-membered furanose ring is drawn in an edgewise projection with the ring oxygen positioned away from the viewer. Ribose, an important aldopentose, commonly adopts a furanose structure, as shown in the following illustration. Cyclic structures of this kind are termed furanose (five-membered) or pyranose (six-membered), reflecting the ring size relationship to the common heterocyclic compounds furan and pyran shown on the right. Five and six-membered rings are favored over other ring sizes because of their low angle and eclipsing strain. If necessary, before you attempt to study this section, review the formation of hemiacetals discussed in Section 19.10.Īs noted above, the preferred structural form of many monosaccharides may be that of a cyclic hemiacetal.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
